Newly published research from MSU scientist unlocks contributors to male fertility
Yuan Wang published her findings in November 2024 in Stem Cell Reports.
EAST LANSING, Mich. — The mitochondrion has long been dubbed as the “powerhouse of the cell,” but there’s still much to learn about its inner workings during development and disease formation. Research recently published by a Michigan State University scientist adds to the understanding of how mitochondria function during spermatogenesis, the process by which sperm cells are produced.
Yuan Wang, an associate professor in the MSU Department of Animal Science, published her findings in November 2024 in Stem Cell Reports.

Wang said roughly 48.5 million couples worldwide are affected by infertility, and about half of the cases are due to male factors.
“The causes of male infertility, and insight into whether any potential treatments for mitochondrial defects may remedy these male infertile cases, are largely unknown,” said Wang, whose work is supported by MSU AgBioResearch.
Mitochondria are highly independent from the rest of the cell. One cell can have hundreds to thousands of mitochondria, and each mitochondrion has its own DNA set separate from the cell, with some having multiple sets.
They’re also responsible for producing most of the cell’s energy in the form of adenosine triphosphate, which is broken down into adenosine diphosphate when energy is needed.
Because mitochondria have such a key role in the cell, knowing how they function in the cells responsible for reproduction is critical. Wang said, however, there are large knowledge gaps in this space that she and her colleagues have been working to fill.
Wang found from studies involving mice that the fusion and fission — the joining together and splitting — of mitochondria are upregulated when spermatogonial differentiation occurs.
What does this mean, exactly?
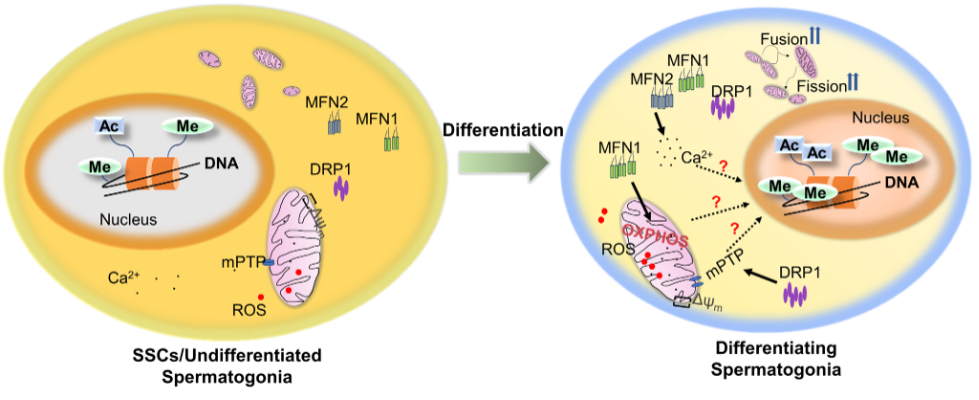
The spermatogonial population in testes, the male reproductive organ that produces sperm and hormones, contain stem cells that have the potential to either self-renew, copy themselves to create more of the same cell, or differentiate, divide themselves to create different cells with specialized functions. In this case, the differentiation of spermatogonial stem cells will ultimately generate sperm for reproduction.
Wang and her team observed that fusion and fission occur at an increased rate in the mitochondria of differentiating spermatogonia, promoting them to become sperm cells.
Proteins, such as the dynamin-related protein 1 (DRP1) and mitofusin 1 and 2 (MFN1 and MFN2), are drivers of mitochondrial fission and fusion. They play an important part in regulating the size, shape and content exchange of mitochondria.
Wang’s team showed that DRP1 upregulates the opening of pores in mitochondria and allows material to be transferred in and out during spermatogonial differentiation, assisting mitochondria in their communication with outside proteins and DNA in the nucleus. Additionally, MFN1 enlarges mitochondria and promotes mitochondrial metabolism to meet the energy demand for differentiating spermatogonia.
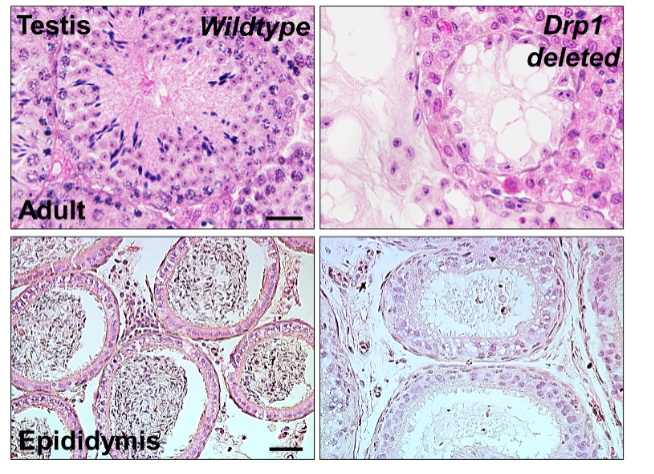
“We found that fusion and fission are very active and also elaborately balanced during spermatogonial differentiation,” Wang said. “If these fusion and fission processes are being disrupted or imbalanced, for example — if energy isn’t being produced or mitochondrial content doesn’t travel in and out properly — spermatogonial differentiation will be blocked and male infertility can take place.”
Knowing this advances reproduction research, Wang said.
Her data reveal if these regulators of mitochondrial fusion and fission don’t perform properly, then spermatogenesis won’t occur, leading to male infertility. These findings improve the understanding behind the mechanistic causes of male infertility due to mitochondrial defects, while providing information on therapeutic targets to improve male fertility impaired by mitochondrial dysfunction.
Funding for this research comes from the National Institutes of Health (NIH). Wang currently leads another NIH-funded project examining the mitochondrial dynamics in female germ cells.
Unlike DNA in the nucleus of the cell where half of it comes from the father and the other half comes from the mother, mitochondria and mitochondrial DNA, as Wang explained, are all inherited from the mother.
“Female cells have hundreds of thousands of mitochondria, compared to a couple hundred in male cells,” Wang said. “Female mitochondria also go to the next generation, so that’s why we think they could have a huge impact on female reproduction and fetal well-being.”
Michigan State University AgBioResearch scientists discover dynamic solutions for food systems and the environment. More than 300 MSU faculty conduct leading-edge research on a variety of topics, from health and climate to agriculture and natural resources. Originally formed in 1888 as the Michigan Agricultural Experiment Station, MSU AgBioResearch oversees numerous on-campus research facilities, as well as 15 outlying centers throughout Michigan. To learn more, visit agbioresearch.msu.edu.



 Print
Print Email
Email




